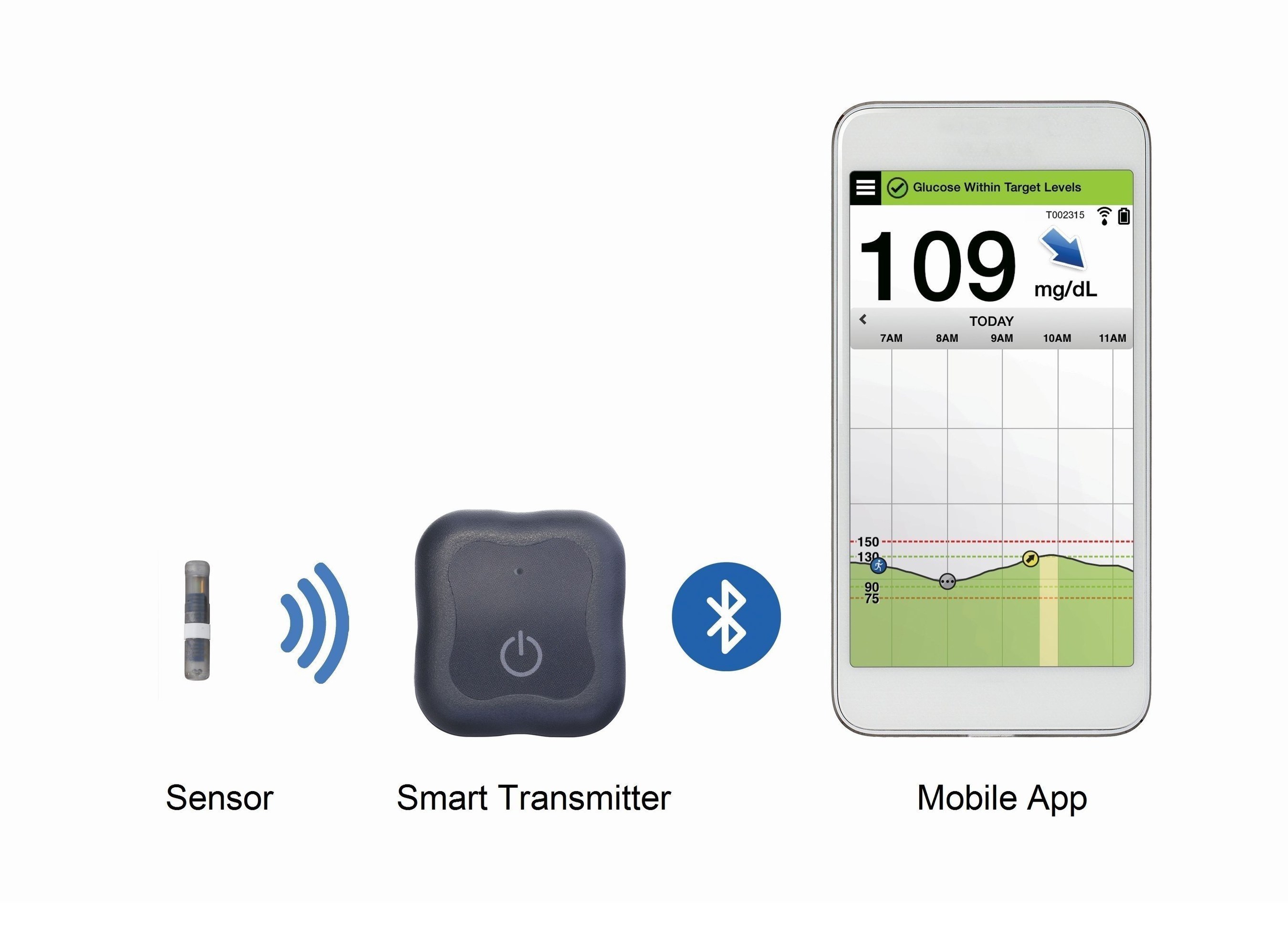
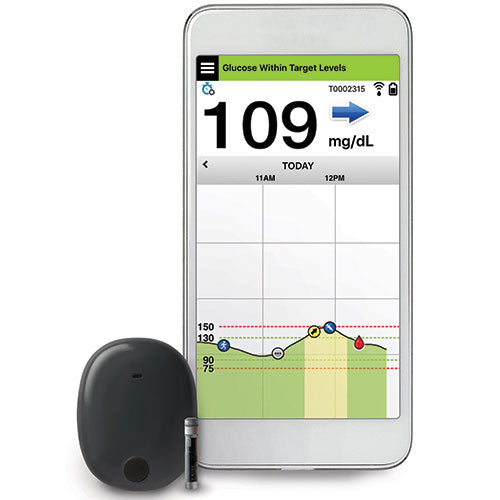
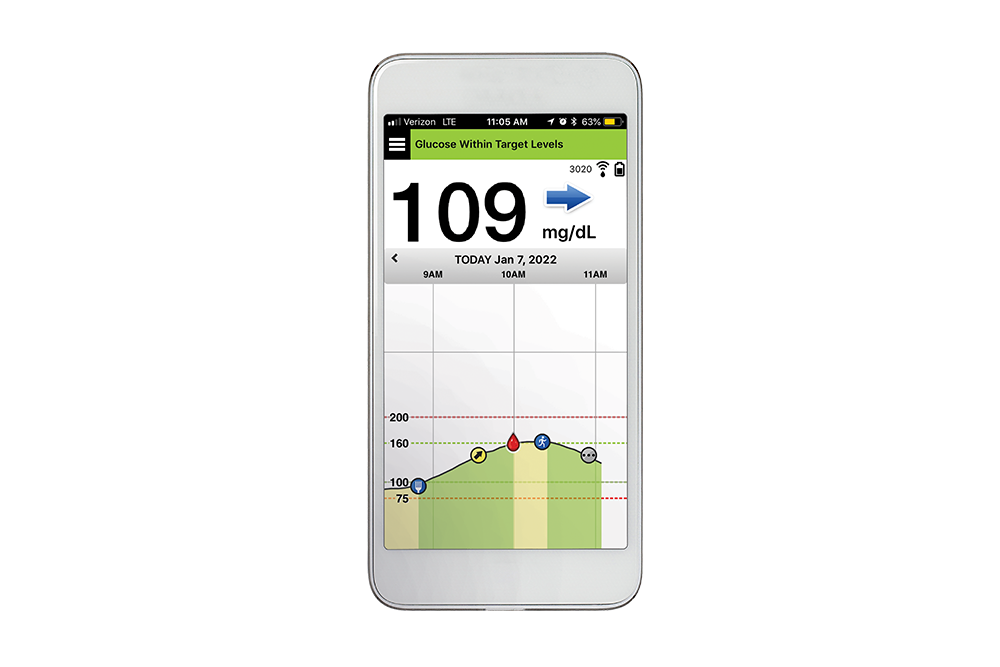
A Prospective Multicenter Evaluation of the Accuracy of a Novel Implanted Continuous Glucose Sensor: PRECISE II | Diabetes Technology & Therapeutics

Eversense sensor. CM, centimeter; DXA, dexamethasone acetate; PMMA,... | Download Scientific Diagram

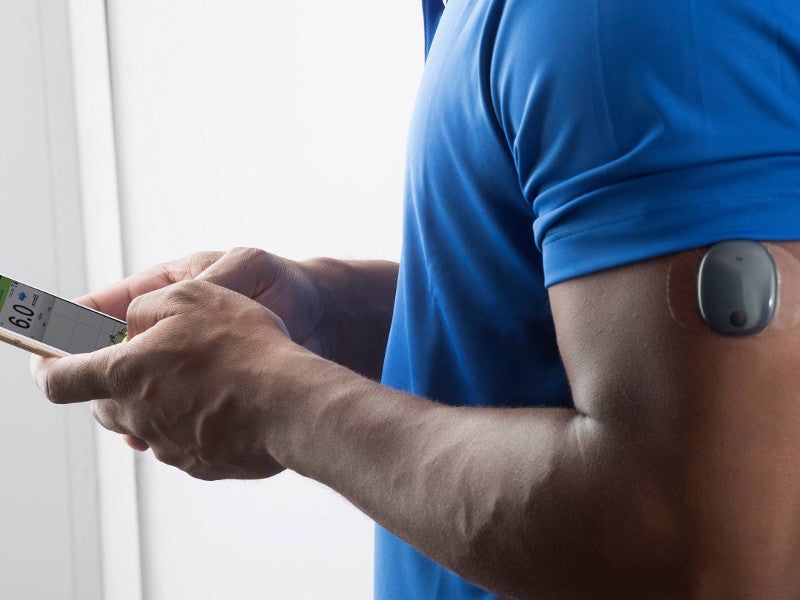
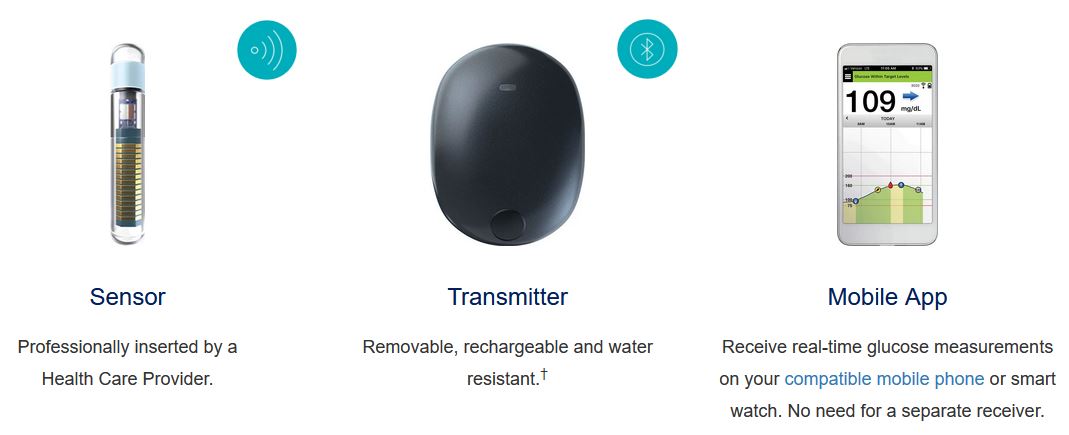
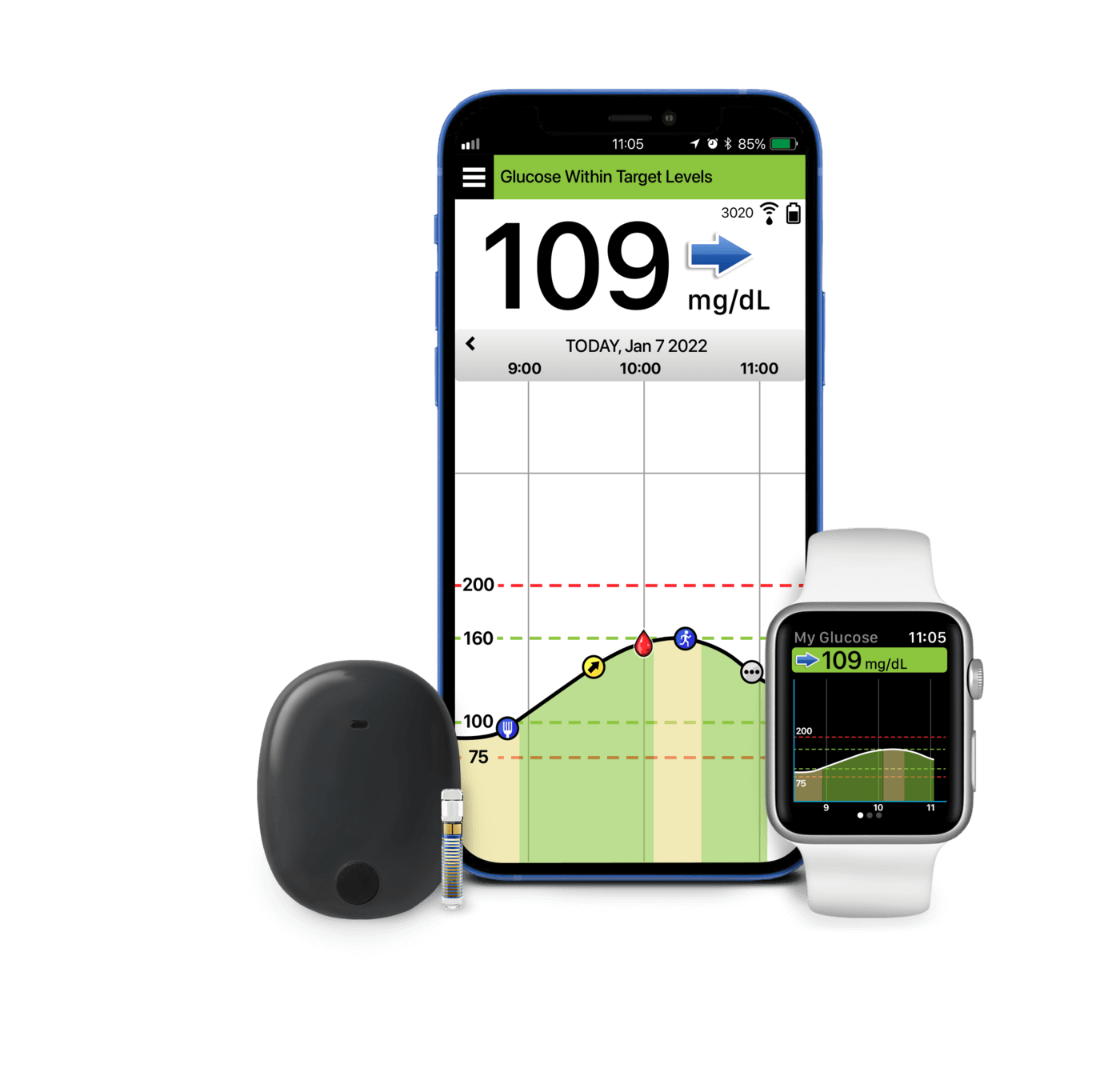
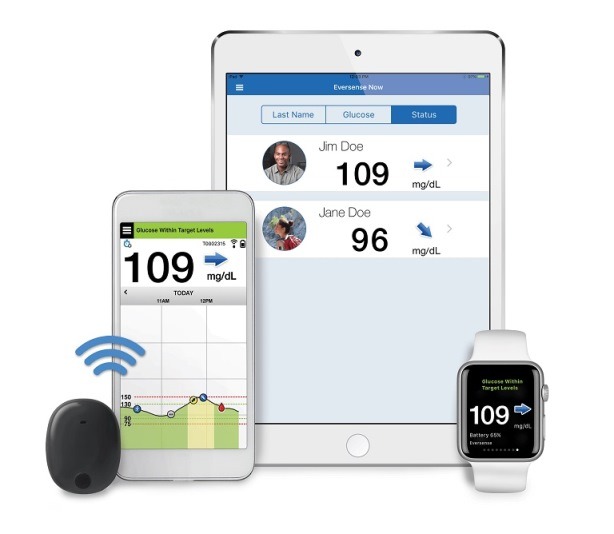
Eversense E3 CGM Approved for Two Sensors per Year: Your “Happily Ever(sense) After” - Taking Control Of Your Diabetes®

Implantable glucose sensor featuring IDT sensing technology awarded CE Mark - Medical Design and Outsourcing

FDA approves Eversense E3 6-month continuous glucose monitor that requires fewer fingerstick blood glucose measurements - NotebookCheck.net News